 
Cations are absorbed by the plant roots and also held on exchange sites in soils. Explanation: Metals lose electrons to nonmetals. 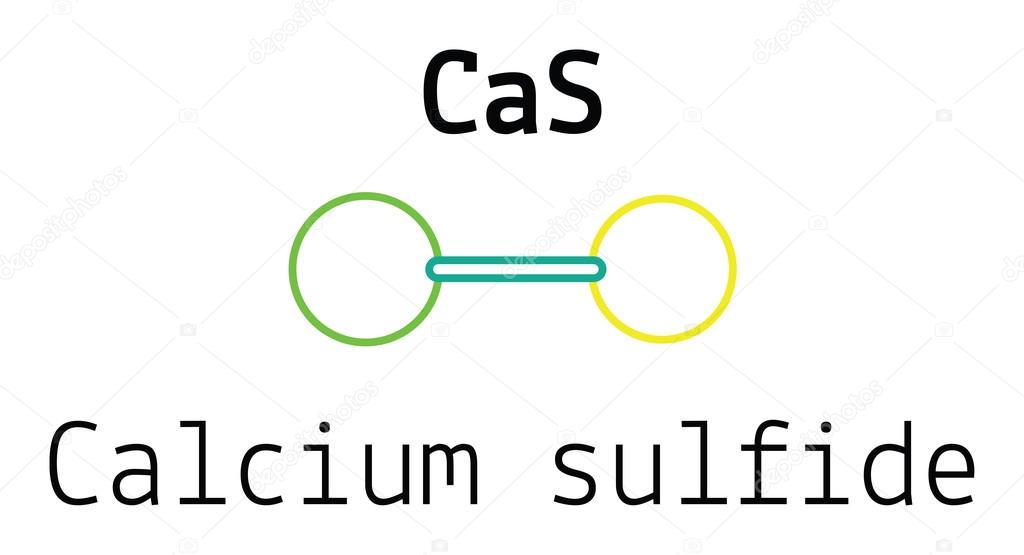 
Because the net ionic charge of a compound is zero, only one of each atom is needed to create calcium sulfide, which will be CaS. = (x=0.5–1), were discovered through a new sol–gel synthetic method that utilizes peroxide ion as a decomposable and stabilizing ligand for titanyl ions in the presence of phosphates in a strong acidic medium. Calcium is a positively charged ion called a cation. The sulfur atom will gain 2 electrons from the calcium atom. Calcium's ion is Ca2+, while the sulfide ion is S2. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
